Regulatory Consulting
Learn how we can help you navigate regulatory requirements, streamline submissions, and ensure your medical device is compliant and submission-ready.
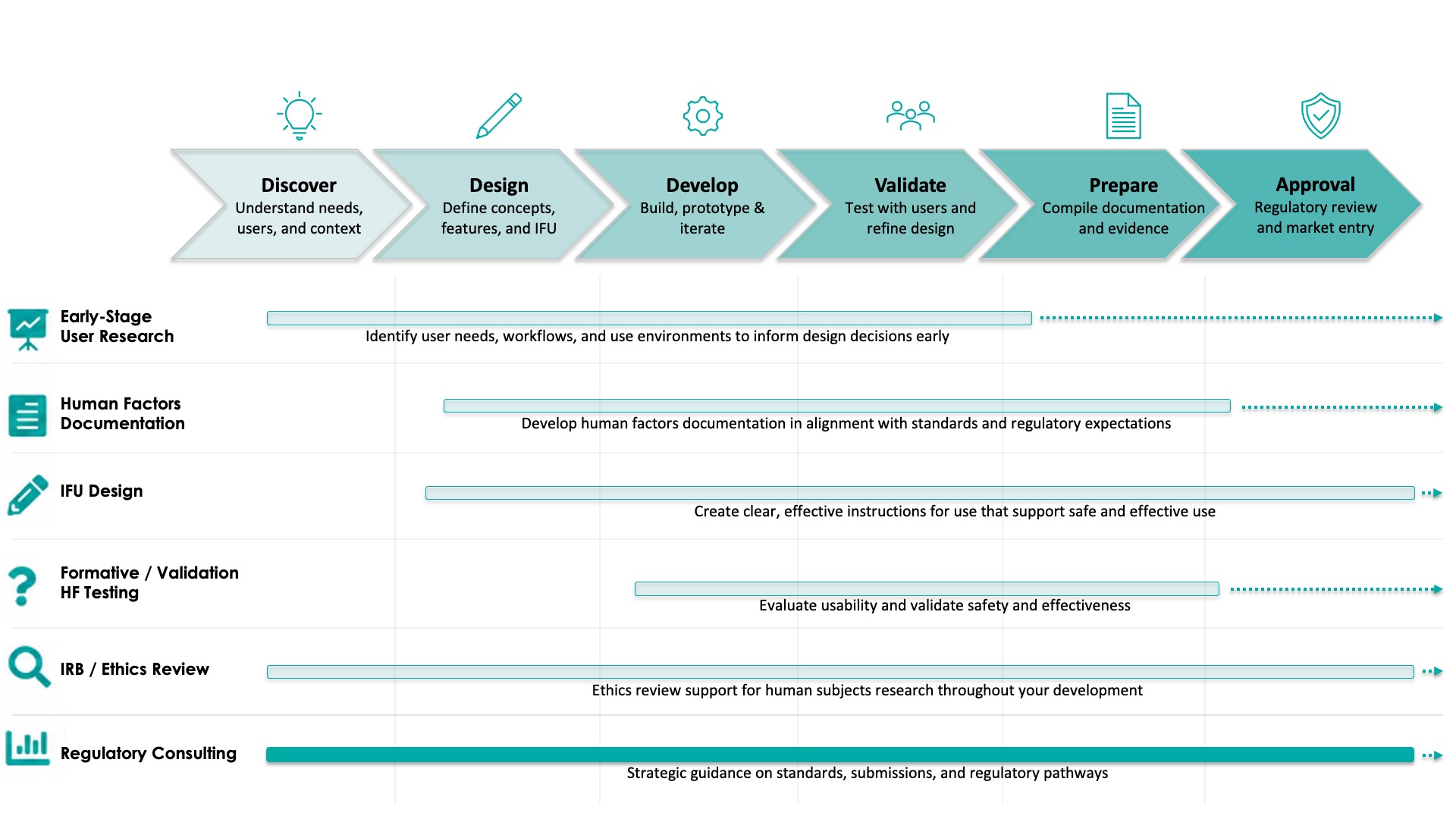
Regulatory consulting plays a critical role in ensuring your human factors work is structured, complete, and aligned with FDA and international expectations.
Strong regulatory strategy helps teams identify gaps in documentation, align with key standards, and prepare for a smooth and efficient submission process.
By engaging regulatory expertise early, organizations can reduce submission risk, avoid delays, and ensure human factors activities are fully aligned with applicable guidance and standards.
Effective regulatory consulting ensures you are meeting regulatory requirements and prepared for successful submission with confidence.
Root Cause Insights provides strategic human factors regulatory consulting, including gap analysis, HFE/UE documentation support, and guidance aligned with FDA and ISO standards.
Strong regulatory strategy helps teams identify gaps in documentation, align with key standards, and prepare for a smooth and efficient submission process.
By engaging regulatory expertise early, organizations can reduce submission risk, avoid delays, and ensure human factors activities are fully aligned with applicable guidance and standards.
Effective regulatory consulting ensures you are meeting regulatory requirements and prepared for successful submission with confidence.
Root Cause Insights provides strategic human factors regulatory consulting, including gap analysis, HFE/UE documentation support, and guidance aligned with FDA and ISO standards.
By engaging regulatory expertise early, organizations can reduce submission risk, avoid delays, and ensure human factors activities are fully aligned with applicable guidance and standards.
Effective regulatory consulting ensures you are meeting regulatory requirements and prepared for successful submission with confidence.
Root Cause Insights provides strategic human factors regulatory consulting, including gap analysis, HFE/UE documentation support, and guidance aligned with FDA and ISO standards.
Effective regulatory consulting ensures you are meeting regulatory requirements and prepared for successful submission with confidence.
Root Cause Insights provides strategic human factors regulatory consulting, including gap analysis, HFE/UE documentation support, and guidance aligned with FDA and ISO standards.
Root Cause Insights provides strategic human factors regulatory consulting, including gap analysis, HFE/UE documentation support, and guidance aligned with FDA and ISO standards.
Our Services
Support regulatory readiness through structured gap analysis, standards alignment, and human factors documentation strategies that strengthen your submission.
HFE / Regulatory Gap Analysis
A structured review of your human factors activities and documentation to identify gaps against FDA guidance and applicable standards. This ensures your submission package is complete, traceable, and aligned with regulatory expectations.
Human Factors Regulatory Strategy
High-level planning support to define the appropriate human factors approach for your device, including study sequencing, documentation needs, and alignment with regulatory pathways. We also support FDA warning letter responses with targeted human factors research data to address identified risks and demonstrate corrective actions.
Standards & Guidance Alignment
Expert interpretation and mapping of key regulatory frameworks including FDA Human Factors Guidance, IEC 62366-1/2, ISO 14971, ISO 13485, and ANSI/AAMI HE75 to your development process and documentation.
Contact Us
Email: info@rootcauseinsights.com
Phone: +1 267-494-9335
Privacy Notice | Terms & Conditions