IRB/Ethics Review
Learn how we protect participants, accelerate study approvals, and keep your research moving forward.
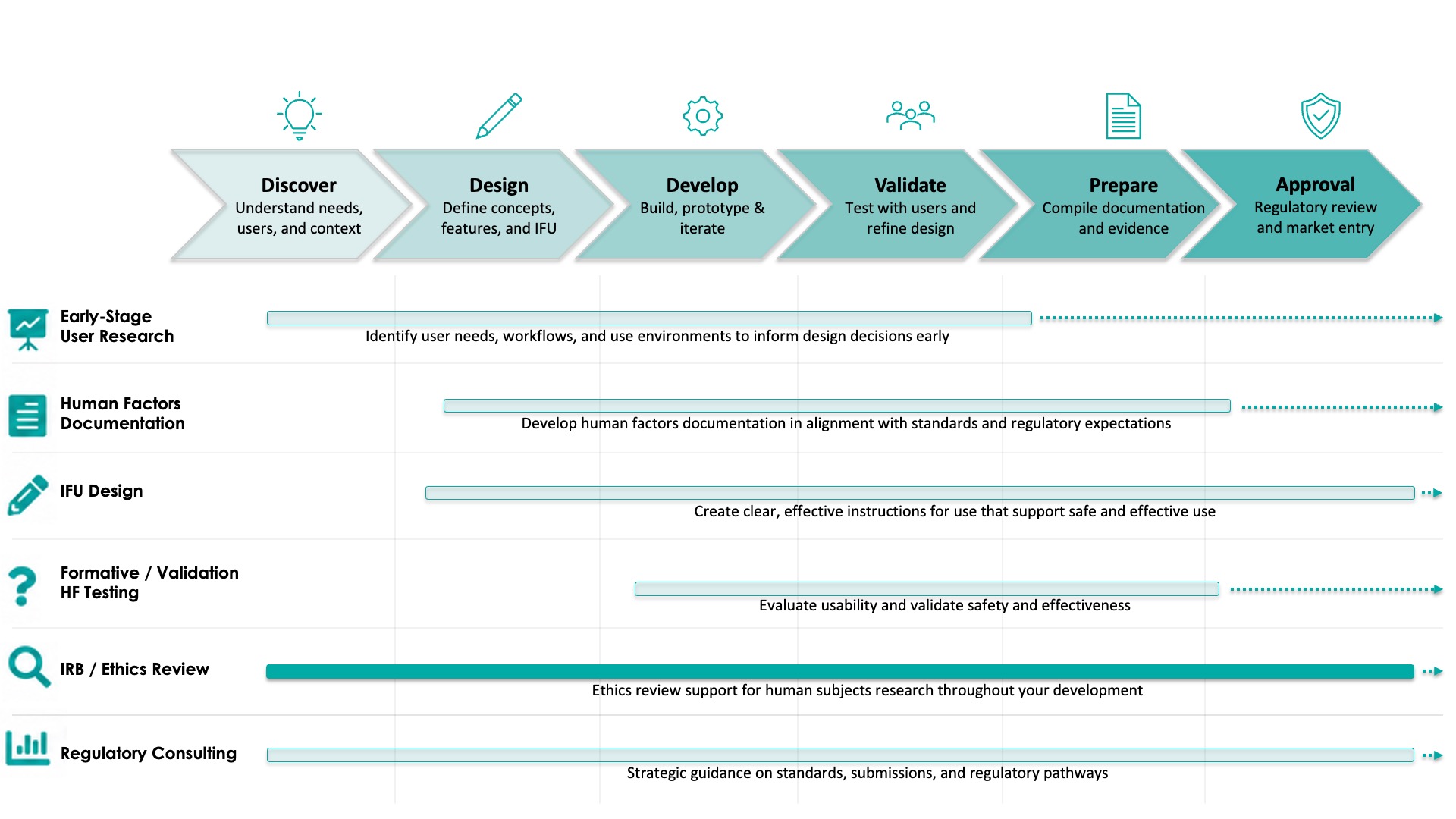
IRB and ethics review is essential for protecting participant rights and ensuring your research meets regulatory requirements.
An efficient review process helps teams move quickly from planning to execution while maintaining compliance with FDA and international guidelines.
By partnering with an experienced IRB, organizations can accelerate approvals, reduce administrative burden, and avoid costly study delays.
Effective ethics review ensures you are protecting participant safety and meeting regulatory expectations with confidence.
Root Cause Insights provides streamlined, responsive IRB and ethics review services to support timely, compliant research execution.
An efficient review process helps teams move quickly from planning to execution while maintaining compliance with FDA and international guidelines.
By partnering with an experienced IRB, organizations can accelerate approvals, reduce administrative burden, and avoid costly study delays.
Effective ethics review ensures you are protecting participant safety and meeting regulatory expectations with confidence.
Root Cause Insights provides streamlined, responsive IRB and ethics review services to support timely, compliant research execution.
By partnering with an experienced IRB, organizations can accelerate approvals, reduce administrative burden, and avoid costly study delays.
Effective ethics review ensures you are protecting participant safety and meeting regulatory expectations with confidence.
Root Cause Insights provides streamlined, responsive IRB and ethics review services to support timely, compliant research execution.
Effective ethics review ensures you are protecting participant safety and meeting regulatory expectations with confidence.
Root Cause Insights provides streamlined, responsive IRB and ethics review services to support timely, compliant research execution.
Root Cause Insights provides streamlined, responsive IRB and ethics review services to support timely, compliant research execution.
Our Services
Streamline IRB and ethics review with efficient, compliant processes that protect participants and accelerate study timelines.
Full IRB Review & Approval
Comprehensive review of study protocols, materials, and methodologies to ensure compliance with FDA regulations and human subject protections. Our IRB has the authority to approve, require modifications, or disapprove studies to support ethical, compliant research.
Recruitment Screener Pre-Review
Early review of recruitment screeners before full protocol submission, allowing participant recruitment to begin sooner. This approach helps maintain timelines while ensuring ethical considerations are addressed from the start.
Global Ethics Review Support
Experience supporting ethics reviews beyond the U.S., including coordination with European bodies such as the UK Health Research Authority (HRA). We help navigate regional requirements to ensure smooth, compliant international research.
Contact Us
Email: info@rootcauseinsights.com
Phone: +1 267-494-9335
Privacy Notice | Terms & Conditions