Human Factors Documentation
Learn how we can help you tell the story of your product’s development and ensure regulatory compliance.
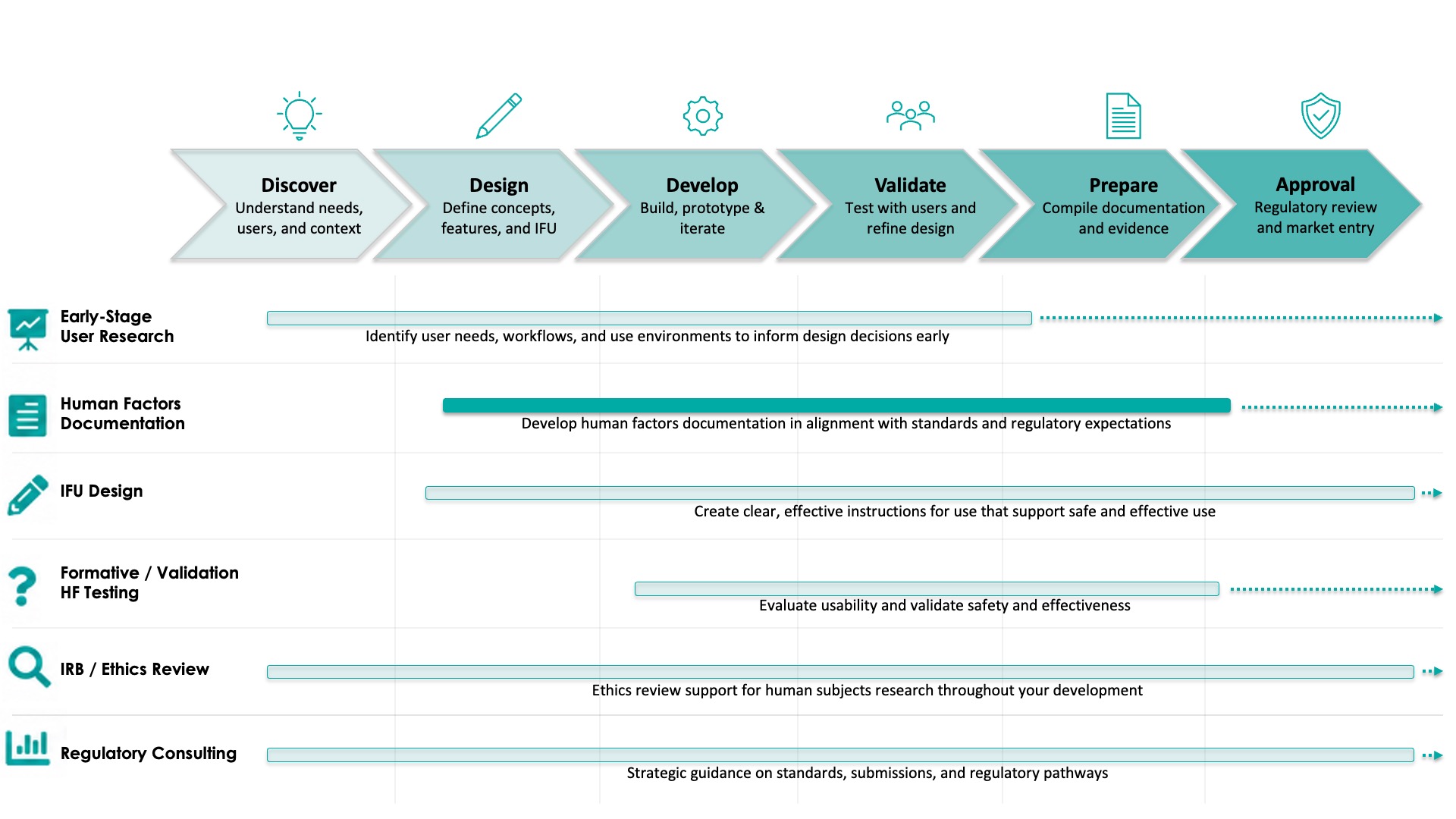
Human Factors documentation is critical for demonstrating that your device is safe, effective, and compliant to FDA standards and guidance.
Comprehensive HFE documentation ensures that user needs, use-related risks, and design decisions are clearly captured and traceable throughout development.
By investing in strong documentation practices, organizations can streamline regulatory submissions, reduce review cycles, and avoid costly delays or rework.
Effective HFE documentation ensures you are demonstrating safe and effective use and meeting regulatory expectations with confidence.
Root Cause Insights partners with teams to develop clear, compliant HFE documentation that supports successful submissions and regulatory approvals.
Comprehensive HFE documentation ensures that user needs, use-related risks, and design decisions are clearly captured and traceable throughout development.
By investing in strong documentation practices, organizations can streamline regulatory submissions, reduce review cycles, and avoid costly delays or rework.
Effective HFE documentation ensures you are demonstrating safe and effective use and meeting regulatory expectations with confidence.
Root Cause Insights partners with teams to develop clear, compliant HFE documentation that supports successful submissions and regulatory approvals.
By investing in strong documentation practices, organizations can streamline regulatory submissions, reduce review cycles, and avoid costly delays or rework.
Effective HFE documentation ensures you are demonstrating safe and effective use and meeting regulatory expectations with confidence.
Root Cause Insights partners with teams to develop clear, compliant HFE documentation that supports successful submissions and regulatory approvals.
Effective HFE documentation ensures you are demonstrating safe and effective use and meeting regulatory expectations with confidence.
Root Cause Insights partners with teams to develop clear, compliant HFE documentation that supports successful submissions and regulatory approvals.
Root Cause Insights partners with teams to develop clear, compliant HFE documentation that supports successful submissions and regulatory approvals.
Our Services
Develop clear, compliant Human Factors documentation that supports regulatory submissions and demonstrates safe and effective use.
HFE / Usability Engineering (UE) Report
A comprehensive document summarizing the full human factors engineering process, including research, risk analysis, formative evaluations, and validation activities. This report demonstrates how use-related risks were identified, mitigated, and validated in alignment with regulatory expectations.
Summative (Validation) Study Report
A detailed report of final validation testing conducted with representative users in simulated use conditions. This document provides evidence that the device can be used safely and effectively, supporting regulatory submissions and demonstrating that critical tasks can be performed without unacceptable risk.
Use-Related Risk Analysis (URRA)
A structured analysis that identifies potential use-related hazards, hazardous situations, and associated risks throughout the user journey. The URRA links user interactions to potential harms and forms the foundation for risk control measures and usability validation activities.
Use Failure Modes and Effects Analysis (UFMEA)
A systematic evaluation of potential user errors and their consequences, including severity and likelihood of occurrence. The UFMEA supports prioritization of design improvements and risk controls, ensuring that critical use-related risks are effectively mitigated.
Contact Us
Email: info@rootcauseinsights.com
Phone: +1 267-494-9335
Privacy Notice | Terms & Conditions